Neuroengineering
Neuroengineering
 Human
Human
 Grade 9+
Grade 9+
Experiment: Wirelessly Control a Cyborg Cockroach
Ever wondered how devices like cochlear implants can use electricity to make people perceive sounds? The answer lies in micro-stimulation of neurons. The RoboRoach lets you study this effect hands-in! Our DIY cyborg experiment lets you briefly influence a cockroach's movements by microstimulating its antenna nerves via Bluetooth backpack. In this lab, you'll learn about microstimulation, surgical prep, animal husbandry, and neuroadaptation!
Experimental procedure
About experiment
What will you learn
- How to perform a simple surgical procedure (antenna implantation) on cockroaches.
- Basics of neural microstimulation and sensory neuron pathways.
- Ethical and scientific considerations when experimenting with living organisms.
- The concept of neural adaptation and learning.
Background
TheRoboRoachis the world’s first commercially available insect cyborg kit—originally designed to teach about advanced neurotechnologies. By sending brief electrical impulses to a roach’s antenna nerves (via a Bluetooth microcontroller backpack), you can evoke turning behaviors. These impulses mimic natural sensory signals, causing the roach to interpret them as obstacles on one side, and briefly turn away. With repeated stimulation, the roach adapts and ignores the stimulus—demonstrating habituation, a fundamental form of learning.
This project is rooted in neural engineering principles used in real-world prosthetics and deep-brain stimulation therapies. The kit includes a small circuit board, a coin battery, and micro-stimulator electrodes. You’ll learn basic cockroach anatomy, how to anesthetize insects safely with ice water, and how to implant electrodes for short-term experiments. We use large discoid cockroaches because they are easy to handle, low in activity, and big enough to carry the backpack. They also have a decentralized nervous system that we can interface with nonlethally.
Experiment

Surgery Preparation
Before starting, gather all necessary supplies: yourRoboRoach kit, a healthy adult cockroach, cold/ice water for anesthesia, sandpaper, super glue gel, hot glue, tweezers, scissors, a small needle, cotton swabs, and a stable workspace. The procedure takes about 1–2 hours, including preparation, surgery, and cleanup.
Important:Only perform the surgery on adult cockroaches (wings visible). Immature roaches still need to molt. Gluing an electrode connector to a juvenile will prevent it from molting properly.
Use ice water to anesthetize the roach. Because cockroaches are ectotherms, cooling them reduces neural activity and renders them temporarily immobile.
Step 1: Secure the Backpack Connector
Anesthetize the roach in ice water (2-5 minutes).
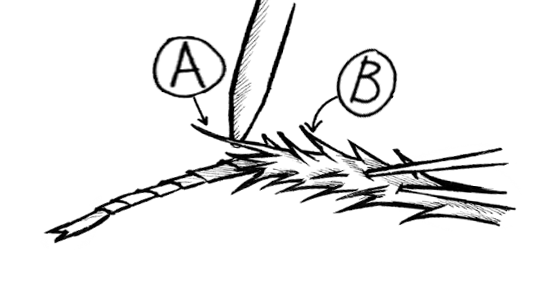
Gently sand the waxy surface (pronotum) on its thorax.
Dab super glue on the sanded area.

Place the black connector so it faces the roach's head. Let dry for ~1 minute.
Return the roach to ice water briefly to maintain anesthesia.

Step 2: Ground Electrode
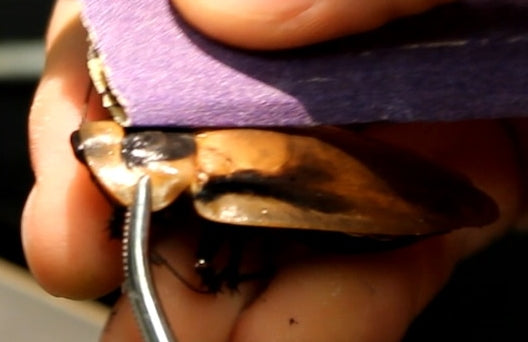
Gently lift a wing to expose the thorax underneath.

Use a small needle to poke a tiny hole in the exoskeleton (off-center).
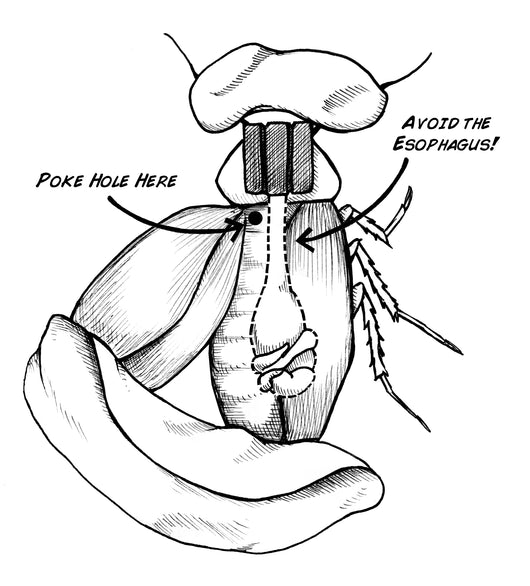
Insert the ground wire ~1–3 mm.
Apply a small bead of super glue around the insertion point. This secures the wire internally.

Step 3: Implanting Antenna Electrodes
Trim ~3–6 mm from each antenna tip.

Insert the left and right silver wires into the corresponding antenna stubs. Secure with a small bead of super glue just inside the cut end.

Ensure minimal slack in the wires. Gather leftover wire on top of the connector and secure with hot glue. Press gently using a floured stick or tool.

Press gently using a floured stick or tool.

Results, Adaptation & Next Steps
Data & Observations:
- Record the time it takes for the cockroach to start ignoring the stimulus—this is habituation.
- Try changing stimulation frequencies and pulse widths. Which settings produce the fastest or slowest turns?
Further Exploration:
- Does adaptation happen faster if the stimuli are more frequent?
- Do some cockroaches adapt more quickly than others?
- How long before the roach regains full sensitivity?
Science Fair Ideas:
- Compare adaptation times vs. stimulation frequency.
- Investigate how temperature affects anesthesia time or recovery.
- Measure at what point the roach stops responding altogether, and how many hours/days pass before it responds again.
References & Resources:
- Backyard Brains Ethics Statement
- American Physiological Society Publications
- NIH Guidelines for Animal Research
- Further reading on cockroach neuroscience and microstimulation
Ethics & Safety: Always handle your cockroach gently. Provide proper care, food, and water. Limit stimulation and allow ample time between trials to minimize stress. Refer to your local guidelines on invertebrate research.
Have fun exploring the frontier of neural interfaces! Share your data and questions with us at hello@backyardbrains.com. Happy experimenting!
What do you need
PRODUCTS AND ACCESSORIES NEEDED: